Did your Boss give you the facts as required by law? Those OSHA laws are 70 years old, and still people argue over excepted facts and tests by NPPTL.
Rember Single use?
I want you to join my group on MeWe or go HERE for misinformation or go to CNN for hoax information.
Are you doing it properly?
Study for OSHA shows Employers in 2020 who had 5A1 Citations for Wilful Neglect of OSHA 29 CFR 1910.134 moved from 4th most common safety violation to 3ed most reported safety violation.
FROM: PATRICK J. KAPUST, Acting Director Directorate of Enforcement Programs 2021 Annual Adjustments to OSHA Civil Penalties
January 8, 2021 U.S. Department of Labor’s OSHA Announces $3,930,381 In Coronavirus Violations OSHA inspections have resulted in the agency citing employers for violations, including failures to:
- Implement a written respiratory protection program;
- Provide a medical evaluation, respirator fit test, training on the proper use of a respirator and personal protective equipment;
- Report an injury, illness or fatality;
- Record an injury or illness on OSHA recordkeeping forms; and
- 5. Comply with the General Duty Clause of the Occupational Safety and Health Act of 1970.
Inspections with COVID-19 related Violations if you look to the far right, you will see hundreds on OSHA 29 CFR 1910.134 5A1 Wilful Neglect violations. In 2020 their was 60,000 plus inspections and this number gets lower every year.
OMG here we have Microfiber Masks that did not pass any of the 510(k) reviews and would fail the NPPTL testing for NIOSH certification for use in hospitals and industries. This KN95, shown with GB2626 2019, is a product of China and those masks failed NIOSH certification because it didn’t allow proper airflow and due to Microfiber being a perfect breeding ground for biological hazards it is not responsible to buy this product, it’s a danger to you and others when not used properly and as a disposable single-use item. https://www.oprahdaily.com/life/health/g38960590/best-kn9product
- “masking is a critical public health tool for preventing the spread of Covid-19,” and “any mask is better than no mask,”
False Both those statements are false. Masks are not at all critical in healthcare, PPE is the least effective form of protection out of all the cross containment prevention measures, and a bad mask can be more dangerous than using a mask, it could already be infected and doesn’t come sterilized**.
- The agency says loosely woven cloth masks provide the least protection, layered finely woven products offer more protection, well-fitting disposable surgical masks and KN95s offer even more protection, and well-fitting NIOSH-approved respirators (including N95s) offer the highest level of protection.
False KN95 and well fitted surgical masks do not offer more protection, especially if their not single-use, their are levels of protection from extensive study, N, R, and P are for Not resistant, water and oil resistant, water and oil protection, as well as 95%, 99%, and 100% filter efficiency, this does not include Bacterial protection efficiency (BPE)
Order of efficiency of FFR’s
Least effective to most effective
SM’s (Surgical Masks)
N95
N99
N100
R95
R99
R100
P95
P99
P100
Half face elastomeric respirator
Air Supplied respirators or PAPR’s
So the best protection is with Air Supplied or PAPR’s
- The N95 and KN95 respirator masks are largely the same, explains Niket Sonpal, MD, an internist in New York City.
False those masks are 100% completely different and KN95’s are a danger to people who already have underlying healtissues like; COPD, athsma, lung cancer or recovering, influenza, Bronchitis, Tuberculosis, Pneumonia, allergies, or when exposed to some chemicals that absorbed into the cloth, as listed in OSHA 29 CFR 1910.134 Appendix (C) with your ANSI/ASSE Z88.2 – 2015 Practices for Respiratory Protection and Z88.6 Respiratory use Qualification exam.
NOTE: Niket Sonpal violated TITLE 18 USC 1038, and is on Oprah’s news website. violation is 5 to 20 years in prison and/or up to $200,000 in fines.
- “Both masks are rated to capture 95 percent of tiny particles (0.3 micron particles),” he says. “The difference between them is considered to be the pressure drop while inhaling, but this is primarily to protect healthcare workers.
FALSE they knew this wouldn’t pass the FDA 510(k) review because it is a negitive pleasure loose-fitting mask, and it’s made out of thick Microfiber (breeding ground for biological hazards) P.S. SM’s and FFR’s like N95’s are tested with 0.1-micron latex spheres not 0.3 as listed on the Fake Oprah’s news website.
- With this in mind, a KN95 mask is your best bet for the most protection against Covid-19 during day-to-day activities.
False masks are the lease effect measures to prevent the spread of biohazards. The most effective is PAPR’s and other Air Supplied Respiratory Protection.
- Finally, adjust the metal nose clip to the shape of your nose and adjust fit as necessary to reduce airflow around the mask.
DANGER necessary to reduce airflow, are you trying to cause hypoxia? Which also is rather deadly and has the same symptoms as Coronavirus, Colds, Flu, SARS, and Pneumonia.
7. Be sure to change out your mask at least every few wears. The CDC says healthcare workers can wear an N95 mask up to five times, says Sonpal.
False it is recommended to be disposable, single-use just as the SM’s and FFR N95’s. It wasn’t ever recommended to use more than once untill.the Pandemic, and their is a list of cleaning agents healthcare can use up to 3 times between patients to conserve on supplies when running low, if it’s not running low it’s single use to prevent spreading biohazards from patient #1 and giving to patient #2 the HAI.
Risks to Health
In the table below, FDA has identified the risks to health generally associated with the use of the surgical masks addressed in this document. The information we recommend you include in your 510(k) to address these identified risks are given in this guidance document, as shown in the table below. We recommend that you conduct a risk analysis, before submitting your 510(k), to identify any other risks specific to your device. The 510(k) should describe the risk analysis method. If you elect to use an alternative approach to address a particular risk identified in this document, or have identified risks additional to those in this document, you should provide sufficient detail to support the approach you have used to address that risk.
Identified risk
Inadequate fluid resistance
Inadequate barrier for bacteria
Inadequate air exchange (differential pressure)
Flammability
Inadequate respiratory barrier for bacteria
NIOSH certification, Surgical masks include parts that have prolonged contact with intact skin. We recommend that you evaluate the biocompatibility of the materials in these parts as described in the standard ISO-10993, “Biological Evaluation of Medical Devices Part 1: Evaluation and Testing” for limited contact devices, contacting intact skin. We also recommend that you document the results in your design history file as a part of the Quality Systems Requirements (21 CFR 820.30). You should select tests appropriate for the duration and level of contact with your device. If identical materials are used in a predicate device with the same type and duration of skin contact, you may identify the predicate device in lieu of performing biocompatibility testing.
“Filtration Efficiency
For surgical masks that are not NIOSH certified N95 Respirators, we recommend that you evaluate filter efficiency performance and bacterial filtration efficiency. For surgical masks that are NIOSH certified N95 Respirators, you may submit your NIOSH certification number in lieu of this information.
Particulate Filtration Efficiency
We recommend that you conduct a particle challenge study using 0.1-Micron Polystyrene Latex Spheres. This in vitro test challenges the mask with unneutralized 0.1-micron polystyrene latex spheres and measures penetration. The use of latex spheres provides an appropriately rigorous test for evaluating a submicron efficiency performance (ASTM F 1215-89 Standard Test Method for Determining the Initial Efficiency of Flatsheet Filter Medium in an Airflow Using Latex Spheres.).
Bacterial Filtration Efficiency
Bacterial Filtration Efficiency (BFE) is a measure of the ability of the mask’s material to prevent the passage of aerosolized bacteria. BFE is expressed in the percentage of a known quantity that does not pass the mask material at a given aerosol flow rate. We recommend that you evaluate the BFE of your device using one of the test methods or standards listed below.
Bacterial Penetration (aerosol filtration) – Mil- M369454C, Military Specifications: Surgical Mask, disposable (June 12, 1975).
Modified Greene and Vesley Method: Method for evaluation of bacterial filtration efficiency of surgical masks. J Bacteriol 83:663-667. (1962).
ASTM F2101-01 Standard Test Method for Evaluating the Bacterial Filtration Efficiency (BFE) of surgical masks using a Biological Aerosol of Staphylococcus aureus.
Back to the top”
“For the U.S. market, Freudenberg Performance Materials is now providing FDA cleared surgical masks meeting the Level 3 standard of the American Society of Testing and Materials (ASTM). The ASTM Level 3 surgical mask is for use in conditions where there is a high risk of fluid and spray of aerosol transmission, such as operating procedures. The masks are a single use, disposable device provided non-sterile“
“Abstract
Respiratory syncytial virus (RSV) infections in hospitalized children were identified by indirect fluorescent antibody technique. Patients with RSV infections were assigned to one of two isolation categories. In one category, the health care workers entering the child’s room did not wear masks and goggles; in the other category, the workers did wear masks and goggles. The RSV illness rate in health care workers using masks and goggles was 5%, but the rate for those using masks and not goggles was 61%. In the no mask/goggles group, the RSV illness rates in the health care workers correlated directly with the number of exposures. In this modest study, the use of masks and goggles was associated with a significant reduction of RSV illnesses in pediatric health care workers.”
https://thumbs.gfycat.com/ClearReflectingEuropeanpolecat-mobile.jpg#h=360w=640
Dr. Fauci lied and many people died
TSA stops enforcing mask mandate on planes after Florida judge ruling
https://thumbs.gfycat.com/LavishFaintHake-size_restricted.gif
“At Denver Children’s Hospital, Murphy et al looked at the effectiveness of gowns and masks in preventing RSV infection in pediatric hospital personnel 6. They found that gowning and masking did not increase personal protection against RSV infection above that which was provided by the usual hand-washing routine 6. We at UCLA and Hall and her colleagues were both aware that conjunctional surfaces could be a site for RSV infections [7–10]. Our group compared standard isolation precautions with standard procedures supplemented by the use of masks and goggles. Among the healthcare workers who wore masks and goggles for clinical care, the infection rate was 5% compared with 61% among the healthcare workers who did not wear masks and goggles “
“Face masks (or medical masks) and respirators are the most commonly used PPE to protect from influenza and other respiratory infection in healthcare settings. However, these two products are not the same. Face masks are not designed for respiratory protection and are used to avoid respiratory droplet and spray of body fluids on the face
VAERS APRIL 8TH 2022
26,976 DEATHS
277 MORE DEATHS IN JUST 7 DAYS
219,865 SERIOUS INJURIES
2,564 MORE INJURIES IN JUST 7 DAYS
MISCARRIAGES 4,496
HEART ATTACKS 13,819
MYO/PERICARDITIS 38,605
PERMANENTLY DISABLED 50,100
LIFE THREATENING 30,293
SHINGLES 13,413
AND THE LIST GOES ON
1.5 hours of Vaccine Adverse Reactions
Who remembers Senator Bill Frist from 2010#1)
Fact references December 20, 2017
27 months later…..March 14, 2020
almost 3 weeks later…..April 3, 2020
https://thumbs.gfycat.com/YellowishTimelyAdder-max-1mb.gif#h=720w=1280
“December 20, 2017 Mr. John Boren 3633 Wareham Drive Thompsons Station, TN 37179
Dear Mr. Boren:
Thank you for your letter to the Occupational Safety and Health Administration (OSHA). Your letter has been referred to the Directorate of Enforcement Programs for an answer to your question. Your letter requested clarification of OSHA’s Respiratory Protection Standard, 29 CFR 1910.134, pertaining to the voluntary use of surgical masks. This letter constitutes OSHA’s interpretation only of the requirements discussed herein, and may not be applicable to any questions not delineated within your original correspondence. Your paraphrased question and our response are below.
Question: Is it permissible to allow surgical masks to be worn on a voluntary basis when respiratory protection is not required to meet any OSHA standards? And if so, is it permissible for employers to provide surgical masks for voluntary use?
Response: Yes. The employer may allow the voluntary use of surgical masks even where an exposure assessment shows respirator use is not required and the employer may provide surgical masks for voluntary use. However, surgical masks may not be used in lieu of required respiratory protection. Surgical masks are not considered respirators by OSHA and, as such, are not covered by 29 CFR 1910.134. They are fluid resistant, disposable, and loose-fitting protection that create a physical barrier between the mouth and nose of the wearer and potential contaminants in the immediate environment. They are commonly used in health care settings for the protection of the patient and they are also often used to prevent splashes from contacting the face of the wearer. However, surgical masks do not seal tightly to the wearer’s face, nor do they provide a reliable level of protection from inhaling smaller airborne particles.
If the hazard to which your employees are exposed to is a combination of splashes and respirable contaminants, your company may want to consider NIOSH approved surgical N95 respirators which also are cleared by the Food and Drug Administration (FDA) for use as a surgical mask. Surgical N95s are filtering facepiece respirators equipped with spray- or splash-resistant facemask material on the outside to protect the wearer from splashes. Regardless of which type is used, the employees should be informed on the different varieties and their unique set of cautions, limitations, and restrictions of use. This information will facilitate employee involvement in the respirator program and/or the overall safety and health program.
For more information on surgical masks and surgical respirators, please review the Hospital Respiratory Protection Program Toolkit at https://www.osha.gov/Publications/OSHA3767.pdf. OSHA also has a fact sheet that compares respirators and surgical masks titled Respiratory Infection Control: Respirators Versus Surgical Masks that is available at https://www.osha.gov/Publications/respirators-vs-surgicalmasks-factsheet.html.
Please be aware that the Tennessee Department of Labor and Workforce Development operates its own occupational safety and health program under an OSHA-approved State Plan. The Tennessee Occupational Safety and Health Administration (TOSHA) adopts and enforces standards and investigates safety and health concerns in workplaces throughout the state. State Plans are required to have standards and an enforcement program that are “at least as effective” as OSHA’s, but may have different or additional requirements. Please contact TOSHA directly at the address below, for further information and to discuss your specific compliance issue:
Tennessee Department of Labor and Workforce Development
220 French Landing Drive
Nashville, Tennessee
Telephone: (615) 741-2793
https://www.tn.gov/workforce/employees/safety-health/tosha.html
Thank you for your interest in occupational safety and health. We hope you find this information helpful. OSHA’s requirements are set by statute, standards, and regulations. Our letters of interpretation do not create new or additional requirements but rather explain these requirements and how they apply to particular circumstances. This letter constitutes OSHA’s interpretation of the requirements discussed. From time to time, letters are affected when the agency updates a standard, a legal decision impacts a standard, or changes in technology affect the interpretation. To assure that you are using the correct information and guidance, please consult OSHA’s website at http://www.osha.gov. If you have further questions, please feel free to contact the Office of Health Enforcement at (202) 693-2190.
Sincerely,
Thomas Galassi, Director
Directorate of Enforcement Programs
Department of Labor logoUNITED”
https://www.osha.gov/laws-regs/standardinterpretations/2017-12-20#:~:text=December%2020%2C%202017,UNITED
https://thumbs.gfycat.com/HandsomeVariableGalapagosalbatross-max-1mb.gif#h=720w=1280
March 14, 2020 MEMORANDUM FOR: REGIONAL ADMINISTRATORS STATE DESIGNEES
THROUGH: AMANDA EDENS, Deputy Assistant Secretary FROM: PATRICK J. KAPUST, Acting Director Directorate of Enforcement Programs
SUBJECT: Temporary Enforcement Guidance – Healthcare Respiratory Protection Annual Fit-Testing for N95 Filtering Facepieces During the COVID-19 Outbreak
This memorandum provides temporary enforcement guidance to Compliance Safety and Health Officers for enforcing the Respiratory Protection standard, 29 CFR § 1910.134, with regard to supply shortages of N95 filtering facepiece respirators due to the COVID-19 outbreak. The Respiratory Protection standard has specific requirements, including a written program, medical evaluation, fit-testing, and training, that employers must follow to ensure workers are provided and are properly using appropriate respiratory protection when necessary to protect their health.1 On March 11, 2020, the President directed the Department of [Health and Human services to “take all appropriate and necessary steps with respect to general use respirators to facilitate their emergency use by healthcare personnel in healthcare facilities and elsewhere,” and he directed the Department of Labor to “consider all appropriate and necessary steps to increase the availability of respirators.”] In light of the Presidential Memorandum, OSHA is providing this temporary guidance for 29 CFR § 1910.134, regarding required annual fit-testing (paragraph (f)(2)), which is to take effect from the date of this memorandum and remain in effect until further notice.
The Centers for Disease Control and Prevention (CDC) currently recommends that Health Care Providers (HCP), who are providing direct care of patients with known or suspected COVID-19, practice infection control procedures. These include engineering controls (e.g., airborne infection isolation rooms), administrative controls (e.g., cohorting patients, designated HCP), work practices (e.g., handwashing, disinfecting surfaces), and appropriate use of personal protective equipment (PPE), such as gloves, face shields or other eye protection, and gowns.
Appropriate respiratory protection is required for all healthcare personnel providing direct care of these patients. For additional guidance, see COVID-19 Hospital Preparedness Assessment Tool, [https://www.cdc.gov/coronavirus/2019-ncov/hcp/hcp-hospital-checklist.html][https_www.cdc.gov_coronavirus_2019-ncov_hcp_hcp-hospital-checklist.html].
OSHA recommends HCP employers follow existing CDC guidelines, including taking measures to conserve supplies of these respirators while safeguarding HCP. One such measure is that healthcare employers may provide HCP with another respirator of equal or higher protection, such as N99 or N100 filtering facepieces, reusable elastomeric respirators with appropriate filters or cartridges, or powered air purifying respirators (PAPR). Another measure is that healthcare employers may change the method of fit testing from a destructive method (i.e., quantitative) to a non-destructive method (i.e., qualitative). For filtering facepiece respirators, qualitative and quantitative fit-testing methods are both effective at determining whether the respirator fits properly. See 29 CFR § 1910.134, Appendix A, [Fit Testing Procedures][].2 The fitted respirator can then be safely used for work tasks that require respiratory protection. For additional guidance, see Strategies for Optimizing the Supply of N95 Respirators, [https://www.cdc.gov/coronavirus/2019-ncov/hcp/respirator-supply-strategies.html][https_www.cdc.gov_coronavirus_2019-ncov_hcp_respirator-supply-strategies.html].3
OSHA field offices shall exercise enforcement discretion concerning the annual fit testing requirement, 29 CFR § 1910.134(f)(2), as long as employers:
Make a good-faith effort to comply with 29 CFR § 1910.134;
Use only NIOSH-certified respirators;
Implement CDC and OSHA strategies for optimizing the supply of N95 filtering facepiece respirators and prioritizing their use, as discussed above;
Perform initial fit tests for each HCP with the same model, style, and size respirator that the worker will be required to wear for protection against COVID-19 (initial fit testing is essential to determine if the respirator properly fits the worker and is capable of providing the expected level of protection);
Inform workers that the employer is temporarily suspending the annual fit testing of N95 filtering facepiece respirators to preserve and prioritize the supply of respirators for use in situations where they are required to be worn;
Explain to workers the importance of performing a user seal check (i.e., a fit check) at each donning to make sure they are getting an adequate seal from their respirator, in accordance with the procedures outlined in 29 CFR § 1910.134, Appendix B-1, User Seal Check Procedures.4 See also, OSHA tutorial videos (English, Spanish).5
Conduct a fit test if they observe visual changes in the employee’s physical condition that could affect respirator fit (e.g., facial scarring, dental changes, cosmetic surgery, or obvious changes in body weight) and explain to workers that, if their face shape has changed since their last fit test, they may no longer be getting a good facial seal with the respirator and, thus, are not being adequately protected; and,
Remind workers that they should inform their supervisor or their respirator program administrator if the integrity and/or fit of their N95 filtering facepiece respirator is compromised.
NOTE: Workers should visually inspect the N95 respirator to determine if the structural and functional integrity of the respirator has been compromised. Over time, components such as the straps, nose bridge, and nose foam material may degrade, which can affect the quality of the fit and seal. If the structural and functional integrity of any part of the respirator is compromised, or if a successful user seal check cannot be performed, discard the respirator and try another respirator.
Where the use of respiratory protection is required and an employer fails to comply with any other requirements, such as initial fit testing, maintenance, care, and training in the Respiratory Protection standard, cite the applicable section(s) of 29 CFR § 1910.134.
To emphasize, this is an enforcement discretion policy, beginning from the date of this memorandum, and applicable where respirators are needed to protect HCP during the COVID-19 outbreak. This temporary enforcement discretion policy will no longer apply upon notification. If you have any questions regarding this policy, please contact Dr. Dionne Williams at (202) 693-2140.
cc: DCSP
DTSEM
DSG
[Correction 4/14/2020]
1 See 29 CFR § 1910.134 at: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.134.
2 See Appendix A, Fit Testing Procedures at: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.134AppA.
3 https://www.cdc.gov/coronavirus/2019-ncov/infection-control/control-recommendations.html.
4 See Appendix B-1, User Seal Check Procedures at: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.134AppB1.
5 See OSHA respirator videos at: https://www.youtube.com/watch?v=Tzpz5fko-fg (English); https://www.youtube.com/watch?v=jgRKuRcTGeE (Spanish).
https://thumbs.gfycat.com/AgreeableSlowBrontosaurus-max-1mb.gif#h=248w=330
April 3, 2020 MEMORANDUM FOR: REGIONAL ADMINISTRATORS STATE PLAN DESIGNEES
THROUGH: AMANDA EDENS, Deputy Assistant Secretary FROM: PATRICK J. KAPUST, Acting Director Directorate of Enforcement Programs
SUBJECT: Enforcement Guidance for Respiratory Protection and the N95 Shortage Due to the Coronavirus Disease 2019 (COVID-19) Pandemic
This memorandum provides interim guidance to Compliance Safety and Health Officers (CSHOs) for enforcing the Respiratory Protection standard, 29 CFR § 1910.134, and certain other health standards, with regard to supply shortages of disposable N95 filtering facepiece respirators. Specifically, it outlines enforcement discretion to permit the extended use and reuse of respirators, as well as the use of respirators that are beyond their manufacturer’s recommended shelf life (sometimes referred to as “expired”). This guidance applies in all industries, including workplaces in which:
Healthcare personnel (HCP) are exposed to patients with suspected or confirmed coronavirus disease 2019 (COVID-19) and other sources of SARS-CoV-2 (the virus that causes COVID-19).
Protection of workers exposed to other respiratory hazards is impacted by the shortage resulting from the response to the COVID-19 pandemic. Such workplace respiratory hazards may be covered by one or more substance-specific health standards.
Our previous memorandum, Temporary Enforcement Guidance – Healthcare Respiratory Protection Annual Fit-Testing for N95 Filtering Facepieces During the COVID-19 Outbreak, issued on March 14, 2020, provided temporary guidance for 29 CFR § 1910.134, regarding required annual fit testing of HCP.1 This memorandum provides additional guidance on enforcing OSHA’s respirator standard for all workers, including HCP. In light of the essential need for adequate supplies of respirators, this memorandum will take effect immediately and remain in effect until further notice. This guidance is intended to be time-limited to the current public health crisis. Please frequently check OSHA’s webpage at www.osha.gov/coronavirus for updates.
Background
The COVID-19 outbreak, which the World Health Organization recently declared a global pandemic, has created an increased demand for N95 filtering facepiece respirators, limiting availability for use in protecting workers in healthcare and emergency response from exposure to the virus. As a result, the President directed the Secretary of Labor to “[consider] all appropriate and necessary steps to increase the availability of respirators.”2
The Food and Drug Administration (FDA) also issued an Emergency Use Authorization (EUA) letter permitting National Institute for Occupational Safety and Health (NIOSH)-approved, disposable filtering facepiece respirators, including those that were NIOSH-approved but have since passed the manufacturer’s recommended shelf life, to be used in healthcare settings to mitigate further transmission of SARS-CoV-2.3
During N95 filtering facepiece respirator (FFR) shortages, the federal government advises that specific N95 FFRs that are beyond their manufacturer’s recommended shelf life will provide greater protection than surgical masks (i.e., facemasks, other than surgical N95s; see below) or non-NIOSH-approved masks (e.g., homemade masks or improvised mouth and nose covers, such as bandanas). NIOSH has tested a sample of N95 FFRs that are beyond their manufacturer’s recommended shelf life from facilities across the United States and determined that certain N95 models continue to protect against the hazards for which they would ordinarily be appropriate (for N95 FFRs, this means they are still expected to filter out 95% of particles of the most penetrating particle size, or 0.3 µm). However, the Centers for Disease Control and Prevention (CDC) and NIOSH have recommended that expired N95 FFRs be used only as outlined in their Strategies for Optimizing the Supply of N95 Respirators.4 For more information, see www.cdc.gov/coronavirus/2019-ncov/release-stockpiled-N95.html.
Enforcement Guidance
All employers whose employees are required to use or are permitted voluntary use of respiratory protection must continue to manage their respiratory protection programs (RPPs) in accordance with the OSHA respirator standard, and should pay close attention to shortages of N95s during the COVID-19 pandemic.5 Paragraph (d)(1)(iii) in section 1910.134 requires such employers to identify and evaluate respiratory hazards in the workplace, and paragraph (c)(1) requires employers to develop and implement written RPPs with worksite-specific procedures and to update their written programs as necessary to reflect changes in workplace conditions that affect respirator use. CSHOs should generally refer to CPL 02-00-158, Inspection Procedures for the Respiratory Protection Standard, 6/26/2014, for further guidance.6
Due to the impact on workplace conditions caused by limited supplies of N95 FFRs, all employers should reassess their engineering controls, work practices, and administrative controls to identify any changes they can make to decrease the need for N95 respirators. Employers should, for example, consider whether it is possible to increase the use of wet methods or portable local exhaust systems or to move operations outdoors. In some instances, an employer may also consider taking steps to temporarily suspend certain non-essential operations.
If respiratory protection must be used, employers may consider use of alternative classes of respirators that provide equal or greater protection compared to an N95 FFR, such as NIOSH-approved, non-disposable, elastomeric respirators or powered, air-purifying respirators (PAPRs). Other filtering facepiece respirators, such as N99, N100, R95, R99, R100, P95, P99, and P100, are also permissible alternatives for those who are unable to obtain N95 FFRs. However, per 29 CFR § 1910.134(d)(1)(ii), when considering N95 alternatives, check to ensure that they are NIOSH-approved, at www.cdc.gov/niosh/npptl/topics/respirators/disp_part/default.html. When these alternatives are not available, or where their use creates additional safety or health hazards, employers may consider the extended use or reuse of N95 FFRs or use of N95 FFRs that were NIOSH-approved but have since passed the manufacturer’s recommended shelf life.
The following specific enforcement guidance is provided for CSHOs inspecting workplaces where workers are using N95 FFRs.
All employers:
Extended use or reuse of N95s:
In the event extended use or reuse of N95 FFRs becomes necessary, the same worker is permitted to extend use of or reuse the respirator, as long as the respirator maintains its structural and functional integrity and the filter material is not physically damaged, soiled, or contaminated (e.g., with blood, oil, paint).7 Employers must address in their written RPPs the circumstances under which a disposable respirator will be considered contaminated and not available for extended use or reuse. Extended use is preferred over reuse due to contact transmission risk associated with donning/doffing during reuse. When respirators are being re-used, employers should pay particular attention to workers’ proper storage of the FFRs in between periods of reuse.
Users should perform a user seal check each time they don a respirator and should not use a respirator on which they cannot perform a successful user seal check. See 29 CFR § 1910.134, Appendix B-1, User Seal Check Procedures.8
Employers should train workers to understand that if the structural and functional integrity of any part of the respirator is compromised, it should be discarded, and that if a successful user seal check cannot be performed, another respirator should be tried to achieve a successful user seal check.
If reuse of respirators is necessary, an appropriate sequence for donning/doffing procedures should be used to prevent contamination, and training needs to address appropriate donning/doffing procedures. See www.cdc.gov/niosh/npptl/pdfs/PPE-Sequence-508.pdf.
Use of expired N95s:
In the event that N95s are not available and the employer has shown a good faith effort to acquire the respirators or to use alternative options, as outlined below, CSHOs should exercise enforcement discretion for the use of N95 FFRs beyond the manufacturer’s recommended shelf life, including surgical N95s.9
Employers may use only previously NIOSH-certified expired N95 FFRs found at www.cdc.gov/coronavirus/2019-ncov/release-stockpiled-N95.html. Workers should be notified that they are using expired N95s.
Purchasers and users of personal protective equipment should not co-mingle products that are past their manufacturer’s recommended shelf life (i.e., expired) with items that are within their shelf life.
Employers should visually inspect, or ensure that workers visually inspect, the N95 FFRs to determine if the structural and functional integrity of the respirator has been compromised. Over time, components such as the straps, nose bridge, and nose foam material may degrade, which can affect the quality of the fit and seal.
Where an employer has expired N95s available from their own stored cache (i.e., not from the U.S. Strategic National Stockpile), the employer should seek assistance from the respirator manufacturer or independent lab regarding testing of those stored respirators prior to use.
Healthcare employers only:
Expired N95s generally must not be used when HCP:
Perform surgical procedures on patients infected with, or potentially infected with, SARS-CoV-2, or perform or are present for procedures expected to generate aerosols or procedures where respiratory secretions are likely to be poorly controlled (e.g., cardiopulmonary resuscitation, intubation, extubation, bronchoscopy, nebulizer therapy, sputum induction).
In accordance with CDC guidance for optimizing the supply of respirators, employers should prioritize the use of N95 respirators by activity type. When HCP perform or are present for aerosol-generating procedures or procedures where respiratory secretions are likely to be poorly controlled, use respirators (including N95 FFRs; other FFRs; non-disposable, elastomeric respirators; and PAPRs) that are still within their manufacturer’s recommended shelf life, if available, before using respirators that are beyond their manufacturer’s recommended shelf life. See www.cdc.gov/coronavirus/2019-ncov/hcp/respirators-strategy/contingency-capacity-strategies.html. The CDC guidance also addresses scenarios in which other crisis standards of care may need to be considered, but this enforcement guidance is not intended to cover those scenarios.
As mentioned above, the FDA has concluded that respirators approved by NIOSH, but not currently meeting the FDA’s requirements, may be protective against SARS-CoV-2. The FDA is providing a list of authorized emergency-use respirators for HCP. Healthcare employers may view the list of approved respirators, and respirator manufacturers and stockpile managers may find information about how to obtain approval for expired respirators, at: www.fda.gov/media/135763/download and http://www.fda.gov/media/135921/download.
Citation guidance:
OSHA will, on a case-by-case basis, exercise enforcement discretion when considering issuing citations under 29 CFR § 1910.134(d) and/or the equivalent respiratory protection provisions of other health standards in cases where:
The employer has made a good faith effort to obtain other alternative filtering facepiece respirators, reusable elastomeric respirators, or PAPRs appropriate to protect workers;
The employer has monitored their supply of N95s and prioritized their use according to CDC guidance (www.cdc.gov/coronavirus/2019-ncov/release-stockpiled-N95.html; www.cdc.gov/coronavirus/2019-ncov/hcp/respirators-strategy/index.html);
Surgical masks and eye protection (e.g., face shields, goggles) were provided as an interim measure to protect against splashes and large droplets (note: surgical masks are not respirators and do not provide protection against aerosol-generating procedures); and
Other feasible measures, such as using partitions, restricting access, cohorting patients (healthcare), or using other engineering controls, work practices, or administrative controls that reduce the need for respiratory protection, were implemented to protect employees.
Where the above efforts are absent and respiratory protection use is required, or voluntary use is permitted, and an employer fails to comply with fit testing, maintenance, care, and training requirements, cite the applicable provision(s) of 29 CFR § 1910.134 and/or other applicable expanded health standards as serious violations. If you have any questions regarding this policy, please contact the Directorate of Enforcement Programs at (202) 693-2190.
cc: DCSP
DTSEM
DSG
[Corrected 4/8/2020]
1 OSHA Memorandum on Temporary Enforcement Guidance – Healthcare Respiratory Protection Annual Fit-Testing for N95 Filtering Facepieces During the COVID-19 Outbreak, March 14, 2020, at www.osha.gov/memos/2020-03-14/temporary-enforcement-guidance-healthcare-respiratory-protection-annual-fit. Back to Text
2 www.whitehouse.gov/presidential-actions/memorandum-making-general-use-respirators-available/. Back to Text
3 www.fda.gov/media/135763/download. Back to Text
4 www.cdc.gov/coronavirus/2019-ncov/hcp/respirator-supply-strategies.html. Back to Text
5 See also, OSHA letter of interpretation to John Boren, Voluntary Use of Surgical Masks, 12/20/2017, at: www.osha.gov/laws-regs/standardinterpretations/2017-12-20. Back to Text
6 www.osha.gov/enforcement/directives/cpl-02-00-158. Back to Text
7 See NIOSH webpage defining the difference between extended use and re-use of N95s at: www.cdc.gov/niosh/topics/hcwcontrols/recommendedguidanceextuse.html. Back to Text
8 User Seal Check Procedures, at: www.osha.gov/laws-regs/ regulations/standardnumber/1910/1910.134AppB1. OSHA tutorial videos are available at: www.youtube.com/watch?v=Tzpz5fko-fg (English); www.youtube.com/watch?v=jgRKuRcTGeE (Spanish). Back to Text
9 Surgical N95s are NIOSH-approved N95 FFRs that are also certified by the FDA for use as a surgical mask. Surgical N95s are normally tested for fluid resistance and flammability. These requirements were not evaluated as part of the NIOSH shelf life testing discussed in this memorandum. CDC/NIOSH does not recommend using N95s beyond the manufacturer-designated shelf life in surgical settings. Back to Text[Back to Text 8]
[Back to Text 8]: https://www.osha.gov/laws-regs/standardinterpretations/2020-04-03#_ftnref9 https://thumbs.gfycat.com/JealousRequiredFennecfox-max-1mb.gif#h=602w=1000
Who remembers Senator Bill Frist from 2010#1)
Fact references December 20, 2017
27 months later…..March 14, 2020
almost 3 weeks later…..April 3, 2020

Even Twitter refuses to allow truth to be posted.
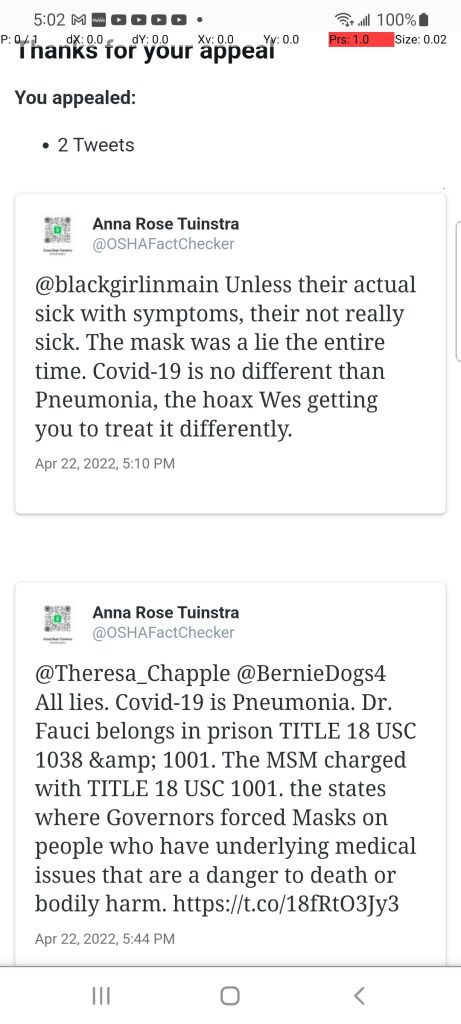
we have 100 plus years of continuous study on Masks.

Do you study history?
Do you study medicine and politics?
Do you have military medical and pandemic Training, vis Submarine training program?
What do you understand about; Emergency Medical Assistance Team, HACCP, OSHA, ServeSafe, HAZMAT, Firefighting, Culinary Specialists, Natropathic enthusiasts, business management, and medical menu planning?
Do you know what a blood bourne diet is to help boost your antigens and immunity via inhibitors?
Do you understand Stewardship’s like; antibody, probiotic, prebiotic, fungal, and parasites?
Let’s look at the Political system for a sec before talking about the masks.
Do the math, more people leave Democrats than Join just as more people join Republicans than leave. Democrats have been around since the begging 246 years ago in 1776 while Republicans started in 1854, 168 years ago. So for a party being 78 years older it sure looks like a dieing Party.
They don’t try to switch, they just do, like Trump and many others [before him](https://en.m.wikipedia.org/wiki/List_of_American_politicians_who_switched_parties_in_office) in fact look at the data, where 138 of our Political leaders elected switched to a different Party.
Leaving Democrats 82 of 138 (59.42%)
80 (57.97%) Democrats switch to Rep Party
1 (0.007%) Democrat switched to Ind Party
1 (0.007%) Democrat switched to Grn Party
Leaving Republican 41 of 138 (28.71%)
28 (20.28%) Republicans switched to Dem Party
9 (0.065%) Republicans switched to Ind Party
2 (0.014%) Republicans switched to Liberal Rep Party
1 (0.007%) Republican switched to Libertarian Party
1 (0.007%) Republican switched to Wisconsin Progressive Party
Joining Democrats 31 of 138 (22.46%)
Joining Republicans 84 of 138 (60.86%)
Notable other 3ed Party
3 (0.021%) Independent switched to Rep Party
2 (0.014%) independent switch to Dem Party
1 (0.007%) Independent switched to Libertarian Party
1 (0.007%) Liberal Rep switched to Rep Party
1 (0.007%) Wisconsin Progressive switched to Rep Party
1 (0.007%) Green switch to no Party reference
1 (0.007%) Reform to Independent Party
This means 4 independent changed to Republicans while only 2 changed to be democratic while 3 stayed independent 3ed Party but a different one of many.
Me, I’m part of the Make Marijuana legal now party.
[](https://oshafactchecker.law.blog/)
#DLM (Dead Lives Mattered) https://thumbs.gfycat.com/DeadWideIrrawaddydolphin-max-1mb.gif#h=368w=640
https://www.c-span.org/video/?190239-1/pandemic-flu-preparedness
December 20, 2017
https://www.osha.gov/laws-regs/standardinterpretations/2017-12-20
March 14, 2020
https://www.osha.gov/laws-regs/standardinterpretations/2020-03-14
April 3, 2020
https://www.osha.gov/laws-regs/standardinterpretations/2020-04-03
https://thumbs.gfycat.com/BoilingTotalIcelandicsheepdog-max-1mb.gif#h=360w=640
End the pandemic hoax by **OSHA FactChecker** aka Matthew Aaron Tuinstra Former Culinary Specialists Navy Submarines and Emergency Medical Assistance Team. Many years of training and instruction for 15 years.
Visit my blog forum expose the plandemic
LikeLiked by 1 person
Copy and paste
**Expose the plandemic**
[](https://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/?preview_id=9&preview_nonce=ef3fae0397&preview=true)%5Bhttps://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/%5D(https://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/?preview_id=9&preview_nonce=ef3fae0397&preview=true)
LikeLiked by 1 person
**Expose the plandemic**
[](https://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/?preview_id=9&preview_nonce=ef3fae0397&preview=true)%5Bhttps://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/%5D(https://oshafactchecker.law.blog/2022/04/18/exposing-the-panldemic/?preview_id=9&preview_nonce=ef3fae0397&preview=true)
Wtf? Democrats are STILL trying to become Republicans new friend. Do the math idiot, more people leave Democrats than Join just as more people join Republicans than leave. Democrats have been around since the begging 296 years ago in 1776 while Republicans started in 1854, 168 years ago. So for a party being 128 years older it sure looks like a dieing Party.
They don’t try to switch, they just do, like Trump and many others [before him](https://en.m.wikipedia.org/wiki/List_of_American_politicians_who_switched_parties_in_office) in fact look at the data, where 138 of our Political leaders elected switched to a different Party.
Leaving Democrats 82 of 138 (59.42%)
80 (57.97%) Democrats switch to Rep Party
1 (0.007%) Democrat switched to Ind Party
1 (0.007%) Democrat switched to Grn Party
Leaving Republican 41 of 138 (28.71%)
28 (20.28%) Republicans switched to Dem Party
9 (0.065%) Republicans switched to Ind Party
2 (0.014%) Republicans switched to Liberal Rep Party
1 (0.007%) Republican switched to Libertarian Party
1 (0.007%) Republican switched to Wisconsin Progressive Party
Joining Democrats 31 of 138 (22.46%)
Joining Republicans 84 of 138 (60.86%)
Notable other 3ed Party
3 (0.021%) Independent switched to Rep Party
2 (0.014%) independent switch to Dem Party
1 (0.007%) Independent switched to Libertarian Party
1 (0.007%) Liberal Rep switched to Rep Party
1 (0.007%) Wisconsin Progressive switched to Rep Party
1 (0.007%) Green switch to no Party reference
1 (0.007%) Reform to Independent Party
Do the math.
Of all the parties
[](https://oshafactchecker.law.blog/)
#DLM (Dead Lives Mattered) https://thumbs.gfycat.com/DeadWideIrrawaddydolphin-max-1mb.gif#h=368w=640
https://www.c-span.org/video/?190239-1/pandemic-flu-preparedness
December 20, 2017
https://www.osha.gov/laws-regs/standardinterpretations/2017-12-20
March 14, 2020
https://www.osha.gov/laws-regs/standardinterpretations/2020-03-14
April 3, 2020
https://www.osha.gov/laws-regs/standardinterpretations/2020-04-03
End the pandemic hoax by OSHA FactChecker
https://thumbs.gfycat.com/BoilingTotalIcelandicsheepdog-max-1mb.gif#h=360w=640
Or go here
I want you to attend my event on MeWe: https://mewe.com/e/smokingajoint
Respiratory Protection for Healthcare Workers Training Video / USDepartmentofLabor
Hey! I’ve been using Cash App to send money and spend using the Cash Card. Try it using my code and we’ll each get $5. XZSMTQT
https://cash.app/app/XZSMTQT
LikeLike
[](https://www.cdc.gov/niosh/npptl/Respiratory-Protection-history.html)
[Personal protective equipment (PPE): PPE ranks lowest in the hierarchy of controls.](https://www.cdc.gov/h1n1flu/guidelines_infection_control.htm#:~:text=Personal%20protective%20equipment%20(PPE)%3A%20PPE%20ranks%20lowest%20in%20the%20hierarchy%20of%20controls.)
[many pregnant women find that they become somewhat shorter of breath as their pregnancy progresses, causing concern that use of N95 FFRs during pregnancy might make breathing even more difficult and possibly harm the woman and her fetus.](https://blogs.cdc.gov/niosh-science-blog/2015/06/18/respirators-pregnancy/#:~:text=many%20pregnant%20women%20find%20that%20they%20become%20somewhat%20shorter%20of%20breath%20as%20their%20pregnancy%20progresses%2C%20causing%20concern%20that%20use%20of%20N95%20FFRs%20during%20pregnancy%20might%20make%20breathing%20even%20more%20difficult%20and%20possibly%20harm%20the%20woman%20and%20her%20fetus)
End the pandemic hoax by OSHA FactChecker https://www.c-span.org/video/?190239-1/pandemic-flu-preparedness
LikeLike
Your YouTube comments can be deleted in 20 second or least, in the video I posted
And it was removed twice.
LikeLike
LikeLike
Learn about those redacted 510k’s for pre-sale, marketing, and sale of. The FDA doesn’t want you knowing the truth about masks and medical device
class 2 & 3.
LikeLike